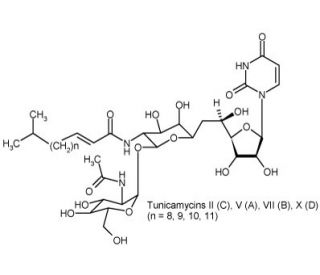
Tunicamycin (CAS 11089-65-9)
See product citations (66)
QUICK LINKS
Tunicamycin is a mixture of tunicamycins A, B, C and D and has been widely used in the study of glycoprotein synthesis in various biological systems. Tunicamycin inhibits GlcNAc phosphotransferase (GPT) and inhibts the formation of N-glycosidic linkages in glycoprotein sunthesis. Tunicamycin has also been reported to have a dose-dependent inhibition of DNA synthesis, inhibit protein glycosylation, suppression of the S phase and to also arrest the cell cycle in late G1. As a member of a family of antibiotics produced by Streptomyces lysosuperficus, tunicamycin is noted to be active in vitro against gram-positive bacteria, fungi, yeasts and viruses. During protein glycosylation, tunicamycin is noted to be an inhibitor of the transfer of saccharide moieties to dolichol during dolichol-linked glycoprotein synthesis. Through this mechanism, it has been postulated to arrest cell cycling as a competitive inhibitor of glycoprotein synthesis. Dose-dependent inhibition of DNA synthesis may be related to the alteration of glycoproteins which thereby affect the transport of thymidine into cells. Additionally, tunicamycin has been reported to prevent cell cycle progression in primary cultures of rat glial cells as well as inhibit lipid-mediated protein glycosylation in chick or mouse fibroblasts in a dose-dependent manner.
Tunicamycin (CAS 11089-65-9) References
- Tunicamycin inhibits capillary endothelial cell proliferation by inducing apoptosis. Targeting dolichol-pathway for generation of new anti-angiogenic therapeutics. | Martínez, JA., et al. 2000. Adv Exp Med Biol. 476: 197-208. PMID: 10949666
- Isoprenoids and astroglial cell cycling: diminished mevalonate availability and inhibition of dolichol-linked glycoprotein synthesis arrest cycling through distinct mechanisms. | Langan, TJ. and Slater, MC. 1991. J Cell Physiol. 149: 284-92. PMID: 1748720
- Endoplasmic reticulum stress induced by tunicamycin and thapsigargin protects against transient ischemic brain injury: Involvement of PARK2-dependent mitophagy. | Zhang, X., et al. 2014. Autophagy. 10: 1801-13. PMID: 25126734
- Tunicamycin enhances the antitumor activity of trastuzumab on breast cancer in vitro and in vivo. | Han, X., et al. 2015. Oncotarget. 6: 38912-25. PMID: 26498681
- GlcNAc-1-P-transferase-tunicamycin complex structure reveals basis for inhibition of N-glycosylation. | Yoo, J., et al. 2018. Nat Struct Mol Biol. 25: 217-224. PMID: 29459785
- Tunicamycin specifically aggravates ER stress and overcomes chemoresistance in multidrug-resistant gastric cancer cells by inhibiting N-glycosylation. | Wu, J., et al. 2018. J Exp Clin Cancer Res. 37: 272. PMID: 30413206
- Tunicamycin induced endoplasmic reticulum changes in endothelial cells investigated in vitro by confocal Raman imaging. | Bik, E., et al. 2019. Analyst. 144: 6561-6569. PMID: 31576836
- Tunicamycin induced endoplasmic reticulum stress in the small intestine. | Öztel, Z., et al. 2021. Biotech Histochem. 96: 507-519. PMID: 32962446
- Tunicamycin-induced endoplasmic reticulum stress inhibits chemoresistance of FaDu hypopharyngeal carcinoma cells in 3D collagen I cultures and in vivo. | Gu, C., et al. 2021. Exp Cell Res. 405: 112725. PMID: 34224701
- Tunicamycin-Induced Endoplasmic Reticulum Stress Promotes Breast Cancer Cell MDA-MB-231 Apoptosis through Inhibiting Wnt/β-Catenin Signaling Pathway. | You, Z., et al. 2021. J Healthc Eng. 2021: 6394514. PMID: 34336161
- The role of GRP78 in oxidative stress induced by tunicamycin in trabecular meshwork cells. | Chai, F., et al. 2022. Acta Biochim Pol. 69: 59-64. PMID: 35233582
- Effect of the endoplasmic reticulum stressor tunicamycin in Angomonas deanei heat-shock protein expression and on the association with the endosymbiotic bacterium. | Catta-Preta, CMC., et al. 2022. Exp Cell Res. 417: 113162. PMID: 35460679
- Pterostilbene Prevents Tunicamycin-Induced Intestinal Barrier Damage by Targeting Endoplasmic Reticulum Stress, Oxidative Stress, Autophagy, and Gut Microbiota. | Chen, Y., et al. 2022. J Agric Food Chem. 70: 13661-13675. PMID: 36225099
- Dolichol-linked glycoprotein synthesis in G1 is necessary for DNA synthesis in synchronized primary cultures of cerebral glia. | Ishii, S. and Volpe, JJ. 1987. J Neurochem. 49: 1606-12. PMID: 3668542
- Relationship of the structure and biological activity of the natural homologues of tunicamycin. | Duksin, D. and Mahoney, WC. 1982. J Biol Chem. 257: 3105-9. PMID: 7061468
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tunicamycin, 5 mg | sc-3506A | 5 mg | $172.00 | |||
Tunicamycin, 10 mg | sc-3506 | 10 mg | $305.00 |
