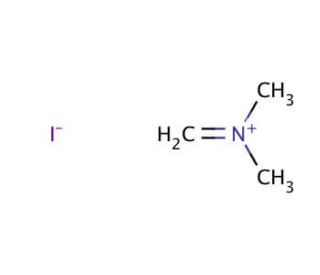
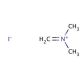
N,N-Dimethylmethyleneiminium iodide (CAS 33797-51-2)
QUICK LINKS
N,N-Dimethylmethyleneammonium iodide (DMMIA) is a versatile quaternary ammonium compound with a distinctive chemical structure, finding extensive applications in scientific research. This colorless salt dissolves readily in water and possesses a melting point of 197°C. N,N-Dimethylmethyleneiminium iodide serves as a valuable reagent for synthesis, facilitating various laboratory experiments. It also contributes to biochemical studies, exhibiting diverse biochemical and physiological effects. N,N-Dimethylmethyleneiminium iodide has proven invaluable in scientific research, serving as a reagent for organic synthesis, a catalyst for alcohol oxidation, and a reagent for the production of heterocyclic compounds. Its utility extends to the synthesis of surfactants and other amphiphilic molecules. Furthermore, N,N-Dimethylmethyleneiminium iodide has been instrumental in investigating enzyme kinetics, as it demonstrates the ability to inhibit certain enzyme activities.
N,N-Dimethylmethyleneiminium iodide (CAS 33797-51-2) References
- Synthetic Routes to Unsymmetrical Porphyrins | Sara Nardis. Synthesis and Modifications of Porphyrinoids. 203–229.
- 2-[(2,3-dihydro-1H-indol-1-yl)methyl]melatonin analogues: a novel class of MT2-selective melatonin receptor antagonists. | Zlotos, DP., et al. 2009. J Med Chem. 52: 826-33. PMID: 19193160
- Synthetic routes to meso-patterned porphyrins. | Lindsey, JS. 2010. Acc Chem Res. 43: 300-11. PMID: 19863076
- Enantioselective approach to quinolizidines: total synthesis of cermizine D and formal syntheses of senepodine G and cermizine C. | Veerasamy, N., et al. 2013. J Org Chem. 78: 4779-800. PMID: 23627426
- High-yielding sequential one-pot synthesis of chiral and achiral α-substituted acrylates via a metal-free reductive coupling reaction. | Ramachary, DB., et al. 2014. Org Biomol Chem. 12: 5400-6. PMID: 24934801
- Synthesis of N-[(dialkylamino)methyl]acrylamides and N-[(dialkylamino)methyl]methacrylamides from Schiff base salts: useful building blocks for smart polymers. | Alzahrani, A., et al. 2018. Org Biomol Chem. 16: 4108-4116. PMID: 29774925
- Indole-based melatonin analogues: Synthetic approaches and biological activity. | Wang, SY., et al. 2020. Eur J Med Chem. 185: 111847. PMID: 31727472
- Unified, Asymmetric Total Synthesis of the Asnovolins and Related Spiromeroterpenoids: A Fragment Coupling Approach. | Yang, F. and Porco, JA. 2022. J Am Chem Soc. 144: 12970-12978. PMID: 35792599
- 1,2-Metallate Rearrangement Using Indole Boronate Species to Access 2,3-Diarylindoles and Indolines. | Das, KK. and Panda, S. 2023. Org Lett. 25: 314-319. PMID: 36602541
- Biomimetic Total Synthesis of Ervitsine and Indole Alkaloids of the Ervatamine Group via 1,4-Dihydropyridines | M.-Lluïsa Bennasar, Bernat Vidal, and Joan Bosch. 1997. J. Org. Chem. 62: 3597–3609.
- Chiral ferrocene derivatives containing a 2,2′-bridged binaphthyl moiety | Michael Widhalm a, Ulrike Nettekoven a, Kurt Mereiter b. 1999. Tetrahedron: Asymmetry. 10: 4369-4391.
- A new method using 2-chloro-4,6-dimethoxy-1,3,5-triazine for facile elimination of dimethylamino group in Eschenmoser's methylenation for synthesis of α,β-unsaturated esters | Kohei Yamada a, Kazumasa Masaki a, Yuri Hagimoto b, Seina Kamiya c, Munetaka Kunishima a b. 2013. Tetrahedron Letters. 54: 1758-1760.
- Functionalization of N,N-Dialkylferrocenesulfonamides toward Substituted Derivatives | Min Wen, William Erb*, Florence Mongin, Yury S. Halauko*, Oleg A. Ivashkevich, Vadim E. Matulis, Thierry Roisnel, and Vincent Dorcet. 2021. Organometallics. 40: 1129–1147.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N-Dimethylmethyleneiminium iodide, 10 g | sc-253145 | 10 g | $102.00 |
