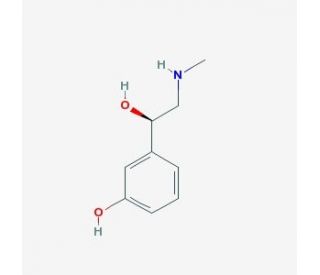
L-phenylephrine (CAS 59-42-7)
See product citations (2)
QUICK LINKS
L-phenylephrine is widely utilized in biochemical research to study its effects on adrenergic receptors, specifically the alpha-1 adrenergic receptor. This compound acts as an agonist at these receptors, which are pivotal in the regulation of smooth muscle tone. Research involving L-phenylephrine often focuses on understanding the signaling pathways activated by these receptors and their role in cellular responses. Additionally, L-phenylephrine is used in studies examining the mechanisms of vasoconstriction and the regulation of blood flow in vascular research. It also serves as a tool in studies related to receptor binding affinity and the specificity of receptor-ligand interactions, providing insights into receptor dynamics and pharmacology.
L-phenylephrine (CAS 59-42-7) References
- Low temperature prevents potentiation of norepinephrine release by phenylephrine. | Santha, E., et al. 2001. Neurochem Int. 38: 237-42. PMID: 11099782
- Dissolution and partitioning behavior of hydrophobic ion-paired compounds. | Lengsfeld, CS., et al. 2002. Pharm Res. 19: 1572-6. PMID: 12425478
- Comparison of the relaxant effects of alfuzosin, phentolamine and sildenafil on rabbit isolated corpus cavernosum. | Palea, S. and Barras, M. 2003. BJU Int. 91: 873-7. PMID: 12780851
- The quantitative measurement of 1-phenylephrine hydrochloride in dilute aqueous solution. | PRATT, EL. 1957. J Am Pharm Assoc Am Pharm Assoc. 46: 505-8. PMID: 13491434
- alpha-1 Adrenergic receptors stimulation induces the proliferation of neural progenitor cells in vitro. | Hiramoto, T., et al. 2006. Neurosci Lett. 408: 25-8. PMID: 16989945
- Diabetic urethropathy compounds the effects of diabetic cystopathy. | Yang, Z., et al. 2007. J Urol. 178: 2213-9. PMID: 17870107
- Epinephrine, phenylephrine, and methoxamine induce infiltrative anesthesia via alpha1-adrenoceptors in rats. | Shieh, JP., et al. 2009. Acta Pharmacol Sin. 30: 1227-36. PMID: 19730427
- Magnesium and diltiazem relaxes phenylephrine-precontracted rat aortic rings. | Dogan, M., et al. 2012. Interact Cardiovasc Thorac Surg. 15: 1-4. PMID: 22523136
- Postsynaptic alpha 1- and alpha 2-adrenoceptors in the circulatory system of the pithed rat: selective stimulation of the alpha 2-type by B-HT 933. | Timmermans, PB. and Van Zwieten, PA. 1980. Eur J Pharmacol. 63: 199-202. PMID: 6247167
- Pharmacological characterization of the postsynaptic alpha adrenoceptors in vascular smooth muscle from canine and rat mesenteric vascular beds. | Agrawal, DK., et al. 1984. J Pharmacol Exp Ther. 229: 831-8. PMID: 6327971
- Antisecretory effects of berberine with morphine, clonidine, L-phenylephrine, yohimbine or neostigmine in pig jejunum. | Zhu, B. and Ahrens, F. 1983. Eur J Pharmacol. 96: 11-9. PMID: 6363101
- Effect of glucagon, phenylephrine, and isoproterenol on glycogenolysis and glucose release from fetal rat hepatocytes in suspension. | Hühn, W., et al. 1983. Biol Neonate. 44: 153-7. PMID: 6626625
- Glutamate inhibition of the adrenergic-stimulated production of melatonin in rat pineal gland in vitro. | Kus, L., et al. 1994. J Neurochem. 62: 2241-5. PMID: 7910630
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-phenylephrine, 5 g | sc-295315 | 5 g | $181.00 | |||
L-phenylephrine, 25 g | sc-295315A | 25 g | $492.00 |
