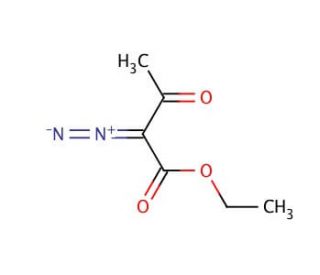
Ethyl diazoacetoacetate (CAS 2009-97-4)
QUICK LINKS
Ethyl diazoacetoacetate proves to be an organic compound with versatile applications in both scientific and industrial domains. It serves as an ester resulting from the combination of butanoic acid and 2-diazo-3-oxobutane. In addition, it has been extensively studied as a promising reagent in organic synthesis, and researchers have explored its potential as a catalyst in specific chemical reactions. Additionally, its attributes as a fluorescent dye have been investigated for potential applications in various imaging techniques. In certain chemical reactions, it is suggested that the compound operates as a proton donor, influencing the reaction′s outcomes. Moreover, the compound′s demonstrated Lewis acid behavior in select organic reactions further enhances its potential as an effective catalyst.
Ethyl diazoacetoacetate (CAS 2009-97-4) References
- A new route to hindered tertiary amines. | Yang, M., et al. 2001. J Org Chem. 66: 6729-33. PMID: 11578227
- Stereoselective synthesis of highly functionalized alpha-diazo-beta-ketoalkanoates via catalytic one-pot Mukaiyama-Aldol reactions. | Zhou, L. and Doyle, MP. 2010. Org Lett. 12: 796-9. PMID: 20102172
- Rh(III)-catalyzed synthesis of multisubstituted isoquinoline and pyridine N-oxides from oximes and diazo compounds. | Shi, Z., et al. 2013. J Am Chem Soc. 135: 12204-7. PMID: 23889167
- Rh(III)-catalyzed synthesis of 1-aminoindole derivatives from 2-acetyl-1-arylhydrazines and diazo compounds in water. | Liang, Y., et al. 2014. Chem Commun (Camb). 50: 6130-3. PMID: 24777073
- Original reactivity of α-diazo-β- ketoesters catalyzed by CpRu complexes. | Tortoreto, C., et al. 2014. Chimia (Aarau). 68: 243-7. PMID: 24983607
- Azirinium ylides from α-diazoketones and 2H-azirines on the route to 2H-1,4-oxazines: three-membered ring opening vs 1,5-cyclization. | Rostovskii, NV., et al. 2015. Beilstein J Org Chem. 11: 302-12. PMID: 25815084
- Regioselective syntheses of 1,2-benzothiazines by rhodium-catalyzed annulation reactions. | Cheng, Y. and Bolm, C. 2015. Angew Chem Int Ed Engl. 54: 12349-52. PMID: 25873036
- Regioselective synthesis of multisubstituted isoquinolones and pyridones via Rh(III)-catalyzed annulation reactions. | Shi, L., et al. 2015. Chem Commun (Camb). 51: 17277-80. PMID: 26463232
- Mechanistic Insight into the Rh(III)-Catalyzed C-H Activation of 2-Acetyl-1-Arythydrazines in Water. | Wu, W., et al. 2017. J Phys Chem A. 121: 1825-1832. PMID: 28182407
- Synthesis of [5,6]-Bicyclic Heterocycles with a Ring-Junction Nitrogen Atom: Rhodium(III)-Catalyzed C-H Functionalization of Alkenyl Azoles. | Halskov, KS., et al. 2017. Angew Chem Int Ed Engl. 56: 9183-9187. PMID: 28586164
- Synthesis of 2,3-Benzodiazepines via Rh(III)-Catalyzed C-H Functionalization of N-Boc Hydrazones with Diazoketoesters. | Wang, J., et al. 2017. Org Lett. 19: 3640-3643. PMID: 28641013
- Cp*Ir(iii)-catalyzed C-H/N-H functionalization of sulfoximines for the synthesis of 1,2-benzothiazines at room temperature. | Aher, YN., et al. 2018. Chem Commun (Camb). 54: 6288-6291. PMID: 29850690
- Theoretical insight into the mechanism, regioselectivity, and substituent group effect of Rh-catalyzed synthesis of 1,2-benzothiazines from NH-sulfoximines and diazo compounds. | Yan, J., et al. 2018. Org Biomol Chem. 16: 5321-5331. PMID: 29999079
- Cp*Ir(III)-Catalyzed C-H/O-H Functionalization of Salicylaldehydes for the Synthesis of Chromones at Room Temperature. | Lade, DM., et al. 2019. J Org Chem. 84: 9188-9195. PMID: 31273978
- Direct Observation of Ylide and Enol Intermediates Formed in Competition with Wolff Rearrangement of Photoexcited Ethyl Diazoacetoacetate. | Phelps, R. and Orr-Ewing, AJ. 2020. J Am Chem Soc. 142: 7836-7844. PMID: 32267699
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl diazoacetoacetate, 1 g | sc-228064 | 1 g | $270.00 |
