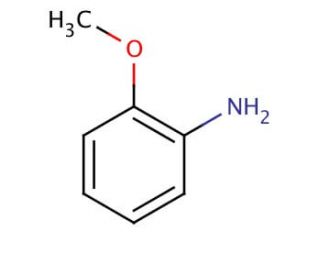
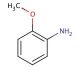
o-Anisidine (CAS 90-04-0)
LINKS RÁPIDOS
A O-Anisidina é um composto de amina aromática da família dos compostos orgânicos. Serve como um precursor vital na síntese de compostos como o fenol, a anilina e o ácido o-toluico. Além disso, a O-Anisidina tem valor como reagente em análises qualitativas e quantitativas em aplicações laboratoriais. O mecanismo de ação da O-Anisidina baseia-se na interação entre os seus átomos de azoto e os electrões de outras moléculas. Os átomos de azoto da O-Anisidina funcionam como nucleófilos, permitindo-lhes reagir com electrófilos. Esta reação química, conhecida como substituição aromática nucleofílica, envolve a deslocação de um átomo de hidrogénio do anel aromático por um nucleófilo. Este padrão de reatividade único contribui ainda mais para a versatilidade do composto e para a sua utilidade em várias investigações científicas.
o-Anisidine (CAS 90-04-0) Referencias
- Os carcinogéneos aminas aromáticas o-toluidina e o-anisidina induzem radicais livres e recombinação intracromossómica em Saccharomyces cerevisiae. | Brennan, RJ. and Schiestl, RH. 1999. Mutat Res. 430: 37-45. PMID: 10592316
- Mecanismo de oxidação mediada pela peroxidase da o-anisidina cancerígena e sua ligação ao ADN. | Stiborová, M., et al. 2002. Mutat Res. 500: 49-66. PMID: 11890934
- Bioensaio do cloridrato de o-anisidina para deteção de possível carcinogenicidade. | ,. 1978. Natl Cancer Inst Carcinog Tech Rep Ser. 89: 1-149. PMID: 12806402
- Identificação de um mecanismo genotóxico para a carcinogenicidade do poluente ambiental e suspeito carcinogéneo humano o-anisidina. | Stiborová, M., et al. 2005. Int J Cancer. 116: 667-78. PMID: 15828049
- Os poluentes cancerígenos o-nitroanisol e o-anisidina são substratos e indutores dos citocromos P450. | Rýdlová, H., et al. 2005. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 149: 441-7. PMID: 16601807
- Identificação de citocromos P450 de rato que metabolizam a N-(2-metoxifenil)hidroxilamina, um metabolito humano dos poluentes ambientais e carcinogéneos o-anisidina e o-nitroanisol. | Naiman, K., et al. 2010. Neuro Endocrinol Lett. 31 Suppl 2: 36-45. PMID: 21187827
- Metabolismo mediado pelo citocromo P450 da N-(2-metoxifenil)-hidroxilamina, um metabolito humano dos poluentes ambientais e carcinogéneos o-anisidina e o-nitroanisol. | Naiman, K., et al. 2008. Interdiscip Toxicol. 1: 218-24. PMID: 21218119
- o-Anisidina e respetivo cloridrato. | ,. 2011. Rep Carcinog. 12: 43-5. PMID: 21829251
- As enzimas do citocromo P450 humano metabolizam a N-(2-metoxifenil)hidroxilamina, um metabolito dos agentes cancerígenos o-anisidina e o-nitroanisol, determinando assim a sua genotoxicidade. | Naiman, K., et al. 2011. Mutat Res. 726: 160-8. PMID: 21946300
- Formação, persistência e identificação de aductos de ADN formados pelo poluente ambiental carcinogénico o-anisidina em ratos. | Naiman, K., et al. 2012. Toxicol Sci. 127: 348-59. PMID: 22403159
- As co-culturas hepáticas in vitro revelam-se adequadas para detetar respostas ao stress oxidativo mediadas por Nrf2 no carcinogéneo da bexiga o-anisidina. | Wewering, F., et al. 2017. Toxicol In Vitro. 40: 153-160. PMID: 28089782
- Diferenças distintas nos mecanismos de lesão da mucosa e formação de γ-H2AX na bexiga urinária de ratos tratados com o-toluidina e o-anisidina. | Toyoda, T., et al. 2019. Arch Toxicol. 93: 753-762. PMID: 30656379
- o-Dímero de Anisidina, 2-Metoxi-N4-(2-metoxifenil) Benzeno-1,4-diamina, na Urina de Rato Associado à Carcinogénese da Bexiga Urinária. | Kobayashi, T., et al. 2021. Chem Res Toxicol. 34: 912-919. PMID: 33587850
- Homo- e Hetero-dímeros Citotóxicos de o-toluidina, o-anisidina e Anilina Formados por Metabolismo In Vitro. | Kobayashi, T., et al. 2022. Chem Res Toxicol. 35: 1625-1630. PMID: 36001821
- Efeitos toxicológicos de dois metabolitos derivados da o-toluidina e da o-anisidina após administração oral de 28 dias a ratos. | Toyoda, T., et al. 2022. J Toxicol Sci. 47: 457-466. PMID: 36328536
Informacoes sobre ordens
| Nome do Produto | Numero de Catalogo | UNID | Preco | Qde | FAVORITOS | |
o-Anisidine, 5 g | sc-250587 | 5 g | $24.00 | |||
o-Anisidine, 100 g | sc-250587A | 100 g | $40.00 |
