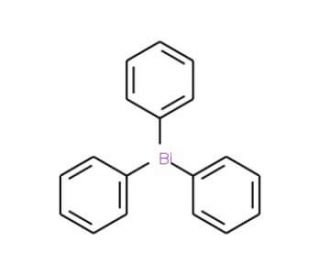
Triphenylbismuth (CAS 603-33-8)
LINK RAPIDI
Il trifenilbismuto è un composto dell'organobismuto che è oggetto di attenzione nel campo della chimica organometallica e della catalisi. Viene utilizzato nella ricerca come catalizzatore o precursore di catalizzatori per varie trasformazioni organiche, comprese le reazioni di formazione del legame C-C, fondamentali nella sintesi di molecole organiche complesse. La sua applicazione nello studio dei processi di polimerizzazione è significativa, in quanto può aiutare a comprendere i meccanismi di innesco e crescita delle catene polimeriche. Nel campo della scienza dei materiali, il trifenilbismuto viene studiato per il suo potenziale utilizzo nella creazione di nuovi materiali con proprietà ottiche o elettroniche uniche, grazie all'effetto dell'atomo pesante del bismuto.
Triphenylbismuth (CAS 603-33-8) Referenze
- L'effetto del trifenilbismuto sulla radiopacità e sulle proprietà prestazionali delle resine per protesi stampate a compressione e a iniezione. | Lang, LA., et al. 2000. J Prosthodont. 9: 23-9. PMID: 11074025
- Effetti citotossici del trifenilbismuto sui timociti di ratto: confronto con il cloruro di bismuto e il cloruro di trifenilstagno. | Arata, T., et al. 2002. Environ Toxicol. 17: 472-7. PMID: 12242678
- Studi strutturali e in soluzione di complessi di fenilbismuto(III) solfonato e loro attività contro l'Helicobacter pylori. | Andrews, PC., et al. 2010. Dalton Trans. 39: 9633-41. PMID: 20830402
- Film sottili di BiFeO3 multiferroici per dispositivi multifunzionali. | Singh, MK., et al. 2010. J Nanosci Nanotechnol. 10: 6195-9. PMID: 21133173
- Sintesi fotochimica di nanocristalli di seleniuro di bismuto in una soluzione micellare acquosa. | Webber, DH. and Brutchey, RL. 2011. Inorg Chem. 50: 723-5. PMID: 21235268
- Effetti relativistici nel trifenilbismuto e loro influenza sulla struttura molecolare e sulle proprietà spettroscopiche. | Berger, RJ., et al. 2012. Phys Chem Chem Phys. 14: 15520-4. PMID: 23072773
- Nuovi complessi di trifenilantimonio(V) e trifenilbismuto(V) con derivati dell'acido benzoico: caratterizzazione strutturale, attività antileishmaniana e antibatterica in vitro e citotossicità contro i macrofagi. | Islam, A., et al. 2014. Molecules. 19: 6009-30. PMID: 24824136
- Effetto del peso molecolare sulle proprietà fisiche degli omopolimeri di poli(etilene brassilato). | Fernández, J., et al. 2016. J Mech Behav Biomed Mater. 64: 209-19. PMID: 27517665
- Superconduttività a 3,5 K e/o 7,2 K nel trifenilbismuto drogato con potassio. | Wang, RS., et al. 2018. J Chem Phys. 149: 144502. PMID: 30316270
- Sintesi, microstruttura e proprietà meccaniche di copoliesteri parzialmente biobased biodegradabili di poli(etilene brassilato-co-ε-caprolattone). | Wei, Z., et al. 2019. J Mech Behav Biomed Mater. 91: 255-265. PMID: 30599448
- Materiali bismuto-organici idrodisperdibili con proprietà di contrasto alla tomografia computerizzata. | Zhang, G., et al. 2018. ACS Appl Bio Mater. 1: 1918-1926. PMID: 31396587
- Trasferimento di carica fotoindotto e dissociazione del legame bimetallico di un complesso Bi-W in soluzione. | Boulanger, SA., et al. 2020. J Phys Chem Lett. 11: 7575-7582. PMID: 32818381
- Caratteristiche di fotoluminescenza e scintillazione di scintillatori plastici a base di PVK bi-caricati per la misurazione ad alta velocità di conteggio di raggi X ad alta energia. | Sato, A., et al. 2021. RSC Adv. 11: 15581-15589. PMID: 35481211
Informazioni ordini
| Nome del prodotto | Codice del prodotto | UNITÀ | Prezzo | Quantità | Preferiti | |
Triphenylbismuth, 5 g (Out of Stock: Availability 4/27/26) | sc-280160 | 5 g | $45.00 | |||
| STATI UNITI: (Esaurito: Disponibilità Aprile 27, 2026) | ||||||
Triphenylbismuth, 25 g | sc-280160A | 25 g | $102.00 | |||
Triphenylbismuth, 100 g | sc-280160B | 100 g | $316.00 | |||
