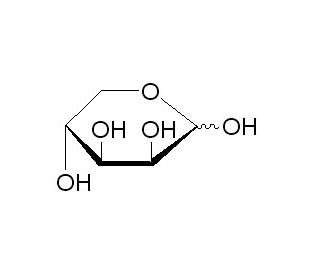
D-(-)-Lyxose (CAS 1114-34-7)
LINK RAPIDI
Il D-(-)-Lixosio è un raro zucchero desossi che si trova naturalmente in piccole quantità in vari organismi. Appartiene alla famiglia dei chetopentosi, un gruppo di monosaccaridi contenenti cinque atomi di carbonio e un gruppo chetonico. La struttura unica di questo zucchero, privo di un atomo di ossigeno rispetto ai suoi omologhi pentosi, lo rende un soggetto interessante per la ricerca biochimica e sintetica. Negli studi scientifici, il D-(-)-Lixosio viene utilizzato per indagare il ruolo e le vie metaboliche di zuccheri rari negli organismi, fornendo approfondimenti sul loro significato biologico e sugli aspetti evolutivi. Le applicazioni di ricerca hanno sfruttato il D-(-)-Lixosio nella sintesi di carboidrati complessi e glicoconiugati grazie alla sua capacità di introdurre diversità strutturale in queste molecole. Ciò ha implicazioni per la comprensione dei processi di riconoscimento e segnalazione biologica mediati dai carboidrati. Inoltre, il D-(-)-Lixosio viene studiato nel contesto delle reazioni enzimatiche che coinvolgono zuccheri rari, favorendo la scoperta e l'ingegnerizzazione di enzimi con nuove funzionalità o specificità. Questi studi contribuiscono a una più ampia comprensione della chimica dei carboidrati e della catalisi enzimatica, enfatizzando l'esplorazione delle vie biochimiche e delle interazioni molecolari che regolano le funzioni cellulari e la diversità biochimica.
D-(-)-Lyxose (CAS 1114-34-7) Referenze
- Valutazione dei protettori dell'analita per migliorare l'analisi gascromatografica dei pesticidi. | Anastassiades, M., et al. 2003. J Chromatogr A. 1015: 163-84. PMID: 14570329
- Analisi degli zuccheri in campioni ambientali mediante gascromatografia-spettrometria di massa. | Medeiros, PM. and Simoneit, BR. 2007. J Chromatogr A. 1141: 271-8. PMID: 17207493
- Impronta metabolica del ceppo LA1147 di Lactobacillus buchneri durante il deterioramento anaerobico di cetrioli fermentati. | Johanningsmeier, SD. and McFeeters, RF. 2015. Int J Food Microbiol. 215: 40-8. PMID: 26325599
- Esplorare lo spazio chimico degli eccipienti per la liofilizzazione. | Meng-Lund, H., et al. 2019. Int J Pharm. 566: 254-263. PMID: 31145963
- Conversione di alcoli in acetali di metilene per reazione con dimetil solfossido-bromo | Munavu, R. M. 1980. The Journal of Organic Chemistry. 45(16): 3341-3343.
- Entalpie di eccesso di soluzioni acquose di monosaccaridi a 298,15 K: pentosi e zuccheri 2-deossi | Barone, G., Castronuovo, G., Doucas, D., Elia, V., & Mattia, C. A. 1983. The Journal of Physical Chemistry. 87(11): 1931-1937.
- Legame selettivo dello zucchero alla β-ciclodestrina: un prototipo per le interazioni zucchero-zucchero in acqua | Aoyama, Y., Nagai, Y., Otsuki, J. I., Kobayashi, K., & Toi, H. 1992. Angewandte Chemie International Edition in English. 31(6): 745-747.
- Analisi qualitativa e quantitativa degli zuccheri caratteristici di tre quinoa colorati basata sulla metabolomica non mirata e mirata | Song, J., & Peng, J. 2024. Journal of Food Composition and Analysis. 126: 105880.
- Caratterizzazione dei principali composti aromatici di un nuovo baijiu Qingke del Tibet mediante GC-MS, GC× GC-MS e GC-O-MS | Zhao, Y., Liao, P., Chen, L., Zhang, Y., Wang, X., Kang, Q.,.. & Sun, J. 2024. Food Chemistry Advances. 4: 100589.
Informazioni ordini
| Nome del prodotto | Codice del prodotto | UNITÀ | Prezzo | Quantità | Preferiti | |
D-(-)-Lyxose, 5 g | sc-221457 | 5 g | $65.00 |
