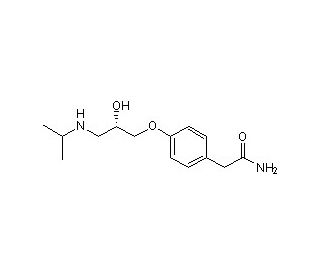
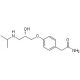
Struttura molecolare di S-(-)-Atenolol, Numero CAS: 93379-54-5
S-(−)-Atenolol (CAS 93379-54-5)
Nomi alternativi:
(S)-(−)-4-[2-Hydroxy-3-[(1-methylethyl)amino]propoxy]benzeneacetamide; (−)-4-[2-Hydroxy-3-[(1-methylethyl)amino]propoxy]benzeneacetamide; Esatenolol
Applicazione:
S-(-)-Atenolol è l'isomero attivo dei preparati racemici di atenololo
Numero CAS:
93379-54-5
Peso molecolare:
266.34
Formula molecolare:
C14H22N2O3
Solo per uso in Ricerca. Non previsto per Uso Diagnostico o Terapeutico.
* Vedere Certificato di Analisi per informazioni sul lotto specifico (incluso il contenuto d'acqua).
LINK RAPIDI
Informazioni ordini
Descrizione
Informazione tecnica
Informazioni di sicurezza
SDS & Certificato d'analisi
L'S-(-)-Atenololo è l'enantiomero attivo dell'atenololo. L'S-(-)-Atenololo è un attivatore dei beta1-AR. L'S-(-)-Atenololo è un antagonista dei recettori beta-adrenergici. Appartiene alla classe dei beta-bloccanti. In laboratorio, questo composto è stato ampiamente utilizzato per esaminare gli effetti cardiovascolari dei beta-bloccanti e per approfondire i meccanismi d'azione di questa classe molecolare. La ricerca sul S-(-)-atenololo comprende studi incentrati sulla comprensione delle differenze di attività tra gli enantiomeri. Questi studi possono fornire indicazioni sulle interazioni composto-recettore e sull'importanza della chiralità.
S-(−)-Atenolol (CAS 93379-54-5) Referenze
- Biotrasformazioni con Rhizopus arrhizus e Geotrichum candidum per la preparazione di (S)-atenololo e (S)-propranololo. | Damle, SV., et al. 2000. Bioorg Med Chem. 8: 2067-70. PMID: 11003151
- Sviluppo e validazione di un metodo cromatografico liquido chirale per la determinazione degli enantiomeri di atenololo e metoprololo in preparati in compresse. | Singh, AK., et al. 2001. J AOAC Int. 84: 1724-9. PMID: 11767137
- Un'efficiente sintesi asimmetrica di (S)-atenololo: utilizzando la risoluzione cinetica idrolitica. | Bose, DS. and Venkat Narsaiah, A. 2005. Bioorg Med Chem. 13: 627-30. PMID: 15653330
- Uso della cromatografia liquida enantioselettiva per la preparazione di enantiomeri puri di atenololo. | Mikuldas, H., et al. 2005. J Sep Sci. 28: 251-6. PMID: 15776927
- Spettroscopia infrarossa di forme racemiche ed enantiomeriche di atenololo. | de Castro, RA., et al. 2007. Spectrochim Acta A Mol Biomol Spectrosc. 67: 1194-200. PMID: 17113823
- Modellazione farmacocinetico-farmacodinamica del S(-)-atenololo nel ratto: riduzione della tachicardia indotta dall'isoprenalina come endpoint farmacodinamico continuo. | van Steeg, TJ., et al. 2007. Br J Pharmacol. 151: 356-66. PMID: 17420778
- Modellazione farmacodinamica basata sul meccanismo del S(-)-atenololo: stima dell'affinità in vivo per il beta1-adrenocettore con un modello di interazione agonista-antagonista. | van Steeg, TJ., et al. 2008. J Pharmacol Exp Ther. 324: 1234-42. PMID: 18162599
- Quantificazione enantioselettiva dell'atenololo nel plasma di topo mediante cromatografia liquida ad alte prestazioni che utilizza una fase stazionaria chirale: applicazione a uno studio farmacocinetico. | Hefnawy, MM., et al. 2013. J AOAC Int. 96: 976-80. PMID: 24282934
- Sintesi di un polimero chirale imprintato di dimensioni nanometriche e suo utilizzo come vettore di (S)-atenololo nella membrana liquida bulk. | Alizadeh, T. 2014. J Sep Sci. 37: 1887-95. PMID: 24771633
- Simulazione di dinamica molecolare e indagine NMR dell'associazione dei β-bloccanti Atenololo e Propranololo con una micella molecolare chirale. | Morris, KF., et al. 2015. Chem Phys. 457: 133-146. PMID: 26257464
- La secrezione renale di atenololo è mediata dal trasportatore 2 di cationi organici umano e dalle proteine di estrusione di farmaci e tossine. | Yin, J., et al. 2015. Drug Metab Dispos. 43: 1872-81. PMID: 26374172
- Separazione e quantificazione di (R)- e (S)-atenololo nel plasma umano e nelle urine utilizzando una colonna di alfa 1-AGP. | Enquist, M. and Hermansson, J. 1989. Chirality. 1: 209-15. PMID: 2642050
- Progettazione, sviluppo e ottimizzazione di compresse a matrice a rilascio prolungato galleggianti di S (-) Atenololo utilizzando la metodologia della risposta superficiale. | Gunjal, PT., et al. 2015. Indian J Pharm Sci. 77: 563-72. PMID: 26798171
- Cromatografia liquida veloce per la separazione dell'atenololo acetato racemico - Il protocollo analitico. | Agustian, J., et al. 2017. Chirality. 29: 847-853. PMID: 28963758
- Farmacocinetica stereoselettiva dell'atenololo nel ratto: influenza dell'invecchiamento e dell'insufficienza renale. | Belpaire, FM., et al. 1993. Mech Ageing Dev. 67: 201-10. PMID: 8469031
Attivatore di:
beta1-AR.Informazioni ordini
| Nome del prodotto | Codice del prodotto | UNITÀ | Prezzo | Quantità | Preferiti | |
S-(−)-Atenolol, 10 mg | sc-203687 | 10 mg | $138.00 | |||
S-(−)-Atenolol, 50 mg | sc-203687A | 50 mg | $357.00 | |||
S-(−)-Atenolol, 2.5 g | sc-203687B | 2.5 g | $2652.00 |
