Heptakis(2,3-dimethyl)-β-cyclodextrin (CAS 123155-05-5)
QUICK LINKS
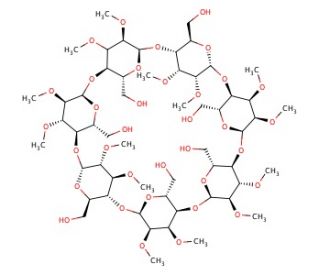
Heptakis(2,3-dimethyl)-β-cyclodextrin is a modified cyclodextrin with seven glucose units in a cyclic arrangement, featuring methyl groups at the 2 and 3 positions of each sugar. This structure alters the inner cavity and hydrophobicity of the molecule, making it a valuable tool in chemical research. Its hydrophobic cavity can encapsulate various hydrophobic compounds, facilitating studies on host-guest chemistry, inclusion complexes, and molecular encapsulation. The methylation enhances solubility in organic solvents and provides greater thermal stability, enabling its use in research involving complexation efficiency under various environmental conditions. Researchers have employed it in analytical chemistry to improve the separation of enantiomers and enhance the performance of chromatographic techniques by stabilizing enantiomeric inclusion complexes. Its role in stabilizing hydrophobic molecules has advanced the understanding of molecular interactions and the influence of substitution patterns on complex formation. Studies using this cyclodextrin derivative have also explored how structural modifications affect binding affinity with various guests, providing insight into designing targeted delivery systems, improving solubilization, and investigating mechanisms of supramolecular recognition. Overall, Heptakis(2,3-dimethyl)-β-cyclodextrin offers a versatile platform for research across analytical chemistry, material science, and molecular recognition.
Heptakis(2,3-dimethyl)-β-cyclodextrin (CAS 123155-05-5) References
- Cyclodextrin-based liquid chromatographic enantiomeric separation of chiral dihydrofurocoumarins, an emerging class of medicinal compounds. | Schumacher, DD., et al. 2003. J Chromatogr A. 1011: 37-47. PMID: 14518761
- Cyclodextrin-based chiral stationary phases for liquid chromatography: a twenty-year overview. | Mitchell, CR. and Armstrong, DW. 2004. Methods Mol Biol. 243: 61-112. PMID: 14970618
- Separation of chiral furan derivatives by liquid chromatography using cyclodextrin-based chiral stationary phases. | Han, X., et al. 2005. J Chromatogr A. 1063: 111-20. PMID: 15700462
- Influence of intramolecular hydrogen bonds on the binding potential of methylated β-cyclodextrin derivatives. | Wenz, G. 2012. Beilstein J Org Chem. 8: 1890-5. PMID: 23209527
- Use of native and derivatized cyclodextrin chiral stationary phases for the enantioseparation of aromatic and aliphatic sulfoxides by high performance liquid chromatography | Mitchell, C., Desai, M., McCulla, R., Jenks, W., & Armstrong, D. 2002. Chromatographia. 56: 127-135.
- Separation of enantiomers of isochromene derivatives by HPLC using cyclodextrin-based stationary phases | Han, X., Zhong, Q., Yue, D., Cà, N. D., Larock, R. C., & Armstrong, D. W. 2005. Chromatographia. 61: 205-211.
- Enantiomeric separation of fused polycycles by HPLC with cyclodextrin and macrocyclic glycopeptide chiral stationary phases | Han, X., Huang, Q., Ding, J., Larock, R. C., & Armstrong, D. W. 2005. Separation science and technology. 40(13): 2745-2759.
- A paper published in Chromatographia1 X. Han, Q. Zhong, D. Yue, N. Della Cà, RC Larock, DW Armstrong | Han, X. 2007. Enantiomeric separations on cyclodextrin-based and synthetic polymeric chiral stationary phases by high performance liquid chromatography and supercritical fluid chromatography. 1001: 46.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Heptakis(2,3-dimethyl)-β-cyclodextrin, 25 mg | sc-396055 | 25 mg | $379.00 |
