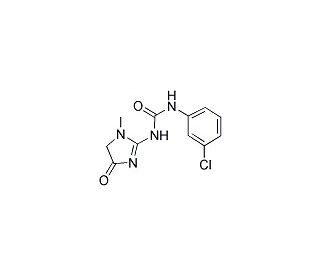
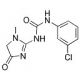
Fenobam (CAS 57653-26-6)
LINK RAPIDI
Il fenobam è un composto che è stato esplorato principalmente per la sua azione come antagonista non competitivo del mGluR5 (recettore metabotropico del glutammato 5). Nella ricerca al di fuori del contesto clinico, è utile negli studi neurologici volti a comprendere la segnalazione glutammatergica e le sue implicazioni per la neuroplasticità e l'eccitotossicità. Le proprietà antagoniste del composto lo hanno reso un punto di riferimento per lo studio della fisiopatologia delle malattie caratterizzate da un'iperattivazione dei recettori del glutammato. Inoltre, Fenobam fornisce un mezzo per studiare la potenziale modulazione della trasmissione sinaptica e i suoi effetti sui circuiti neuronali, che è fondamentale per lo sviluppo di nuove strategie di studio delle funzioni cerebrali. Il suo utilizzo in modelli di ricerca aiuta a decifrare i ruoli complessi dei recettori mGluR5 in vari aspetti della cognizione e del comportamento.
Fenobam (CAS 57653-26-6) Referenze
- Fenobam: un ansiolitico non benzodiazepinico clinicamente validato, è un antagonista potente, selettivo e non competitivo dei recettori mGlu5 con attività agonista inversa. | Porter, RH., et al. 2005. J Pharmacol Exp Ther. 315: 711-21. PMID: 16040814
- Fenil uree della creatinina come antagonisti del mGluR5. Uno studio di relazione struttura-attività sugli analoghi del fenobam. | Wållberg, A., et al. 2006. Bioorg Med Chem Lett. 16: 1142-5. PMID: 16380255
- Le proprietà ansiolitiche e analgesiche del fenobam, un potente antagonista del recettore mGlu5, in relazione alla compromissione dell'apprendimento. | Jacob, W., et al. 2009. Neuropharmacology. 57: 97-108. PMID: 19426746
- Un antagonista mGluR5 in fase di sviluppo clinico migliora la discinesia indotta dalla L-DOPA in ratti e scimmie parkinsoniani. | Rylander, D., et al. 2010. Neurobiol Dis. 39: 352-61. PMID: 20452425
- Antagonismo del recettore metabotropico del glutammato 5 con fenobam: esame della tolleranza analgesica e del profilo degli effetti collaterali nei topi. | Montana, MC., et al. 2011. Anesthesiology. 115: 1239-50. PMID: 22037639
- Il fenobam ha promosso l'effetto neuroprotettivo della PEP-1-FK506BP in seguito a stress ossidativo, aumentandone l'efficienza di trasduzione. | Ahn, EH., et al. 2013. BMB Rep. 46: 561-6. PMID: 24152913
- Il trattamento combinato con fenobam e amantadina promuove robusti effetti antidiscinetici nel modello di primate della malattia di Parkinson con 1-metil-4-fenil-1,2,3,6-tetraidropiridina (MPTP). | Ko, WK., et al. 2014. Mov Disord. 29: 772-9. PMID: 24610195
- L'antagonista mGluR5 fenobam induce una preferenza analgesica condizionata per il luogo in topi con lesioni ai nervi spinali. | Lax, NC., et al. 2014. PLoS One. 9: e103524. PMID: 25061818
- Effetti dei modulatori allosterici positivi e negativi di mGluR5 sul danno cerebrale evocato dall'ipossia-ischemia nei ratti neonati. | Makarewicz, D., et al. 2015. Folia Neuropathol. 53: 301-8. PMID: 26785364
- Docking, simulazione di dinamica molecolare e sintesi di nuovi analoghi di fenobam come antagonisti del recettore mGlu5. | Javidan, A., et al. 2016. Comb Chem High Throughput Screen. 19: 764-770. PMID: 27585831
- I composti pro-psicotici dei recettori metabotropici del glutammato, fenobam e AZD9272, condividono i siti di legame con gli inibitori della monoamino ossidasi-B nell'uomo. | Varnäs, K., et al. 2020. Neuropharmacology. 162: 107809. PMID: 31589885
- Determinazione della preferenza tautomerica del fenobam in soluzione mediante spettroscopia NMR ad alta risoluzione. | Sha, X., et al. 2021. Magn Reson Chem. 59: 641-647. PMID: 33368586
- Il DMSO ritarda la paralisi indotta dalla malattia di Alzheimer in C. elegans attraverso la modulazione della neurotrasmissione di glutammato e acetilcolina. | Sadananda, G., et al. 2021. Ann Neurosci. 28: 55-64. PMID: 34733055
- Nuovi ansiolitici non-benzodiazepinici. | Goldberg, ME., et al. 1983. Neuropharmacology. 22: 1499-504. PMID: 6142427
- Metabolismo in vitro e in vivo dell'agente ansiolitico fenobam nel ratto. | Wu, WN., et al. 1995. J Pharm Sci. 84: 185-9. PMID: 7738798
Informazioni ordini
| Nome del prodotto | Codice del prodotto | UNITÀ | Prezzo | Quantità | Preferiti | |
Fenobam, 5 mg | sc-202608 | 5 mg | $86.00 | |||
Fenobam, 25 mg | sc-202608A | 25 mg | $306.00 |
