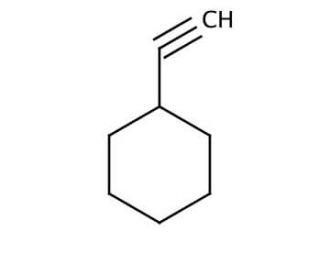
Cyclohexylacetylene (CAS 931-48-6)
QUICK LINKS
Cyclohexylacetylene (CHA) is an important organic compound that consists of a cyclohexyl group attached to an acetylene group. The unique properties of Cyclohexylacetylene include its ability to form diverse cyclic and linear polymers. Moreover, Cyclohexylacetylene has proven to be an effective catalyst in a wide range of reactions, including the polymerization of olefins and the synthesis of polymers. The versatility of cyclohexylacetylene has led to its application in various scientific research endeavors. It has been employed in the synthesis of polymers and catalysts, facilitating advancements in these fields. While the precise mechanism of action of cyclohexylacetylene is not fully elucidated, it is believed to involve the formation of a cyclic polyene. This cyclic polyene is thought to interact with a range of molecules, including proteins, enzymes, and other organic compounds.
Cyclohexylacetylene (CAS 931-48-6) References
- Traceless Rhodium-Catalyzed Hydroacylation Using Alkyl Aldehydes: The Enantioselective Synthesis of β-Aryl Ketones. | Bouisseau, A., et al. 2016. Chemistry. 22: 15624-15628. PMID: 27666437
- Efficient Palladium-Catalyzed Aerobic Arylative Carbocyclization of Enallenynes. | Liu, J., et al. 2018. Angew Chem Int Ed Engl. 57: 16842-16846. PMID: 30351460
- Vinyl Cation Stabilization by Silicon Enables a Formal Metal-Free α-Arylation of Alkyl Ketones. | Pons, A., et al. 2019. Angew Chem Int Ed Engl. 58: 17303-17306. PMID: 31638738
- Ligand-Controlled Diastereoselective Cobalt-Catalysed Hydroalkynylation of Terminal Alkynes to E- or Z-1,3-Enynes. | Weber, SM., et al. 2020. Chemistry. 26: 12129-12133. PMID: 32271963
- Efficient [(NHC)Au(NTf2)]-catalyzed hydrohydrazidation of terminal and internal alkynes. | Heidrich, M. and Plenio, H. 2020. Beilstein J Org Chem. 16: 2080-2086. PMID: 32952724
- Highly regio- and stereoselective phosphinylphosphination of terminal alkynes with tetraphenyldiphosphine monoxide under radical conditions. | Tran, DP., et al. 2021. Beilstein J Org Chem. 17: 866-872. PMID: 33968259
- Decarbonylative Sonogashira Cross-Coupling of Carboxylic Acids. | Liu, C. and Szostak, M. 2021. Org Lett. 23: 4726-4730. PMID: 34096312
- Palladium-Catalyzed Cascade Carbonylation to α,β-Unsaturated Piperidones via Selective Cleavage of Carbon-Carbon Triple Bonds. | Ge, Y., et al. 2021. Angew Chem Int Ed Engl. 60: 22393-22400. PMID: 34382728
- Palladium-Catalyzed Domino Aminocarbonylation of Alkynols: Direct and Selective Synthesis of Itaconimides. | Ge, Y., et al. 2021. JACS Au. 1: 1257-1265. PMID: 34467363
- Decreasing the coordinated N atoms in a single-atom Cu catalyst to achieve selective transfer hydrogenation of alkynes. | Zhang, X., et al. 2021. Chem Sci. 12: 14599-14605. PMID: 34881012
- Three-Component Friedel-Crafts Transformations: Synthesis of Alkyl and Alkenyl Trifluoromethyl Sulfides and Alkenyl Iodides. | Chu, D. and Ellman, JA. 2022. Org Lett. 24: 2921-2925. PMID: 35394794
- Synergistic effects of core@double-shell structured magnesium hydroxide microcapsules on flame retardancy and smoke suppression in flexible poly(vinyl chloride). | Xu, J., et al. 2022. RSC Adv. 12: 2914-2927. PMID: 35425324
- Parts-Per-Million of Soluble Pd0 Catalyze the Semi-Hydrogenation Reaction of Alkynes to Alkenes. | Ballesteros-Soberanas, J., et al. 2023. J Org Chem. 88: 18-26. PMID: 35584367
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cyclohexylacetylene, 1 g | sc-234455 | 1 g | $57.00 |
