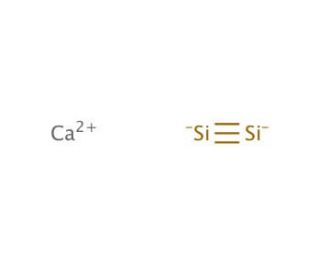
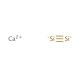
Calcium silicide (CAS 12013-56-8)
Direktverknüpfungen
Calciumsilicid ist eine Verbindung, die in metallurgischen Prozessen als starkes Desoxidations- und Entschwefelungsmittel wirkt. Wird es geschmolzenem Stahl oder Eisen zugesetzt, reagiert es mit Sauerstoff und Schwefel und bildet Calciumoxid und Calciumsulfid. Diese Reaktion trägt dazu bei, Verunreinigungen aus dem Metall zu entfernen, was zu einem saubereren und feineren Endprodukt führt. Calciumsilicid wird als Keimbildner bei der Herstellung von Gusseisen verwendet, wo es die Bildung von Graphitflocken fördert und so die mechanischen Eigenschaften des Materials verbessert. Auf molekularer Ebene interagiert Calciumsilicid mit den im Metall vorhandenen Verunreinigungen und bildet stabile Verbindungen, die sich leicht entfernen lassen, wodurch die Qualität des Metalls verbessert wird. Seine Wirkungsweise besteht in der Bildung stabiler Verbindungen mit Sauerstoff und Schwefel, was zu deren Entfernung aus dem geschmolzenen Metall führt und letztlich zur Herstellung hochwertiger Stahl- und Eisenprodukte beiträgt.
Calcium silicide (CAS 12013-56-8) Literaturhinweise
- Auslaugung von Aluminium und Eisen aus Kesselschlacke, die in einem typischen chinesischen Stahlwerk anfällt. | Li, J., et al. 2009. J Hazard Mater. 166: 1096-101. PMID: 19157693
- Chemische und physikalische Charakterisierung von Schweißrauchpartikeln zur Unterscheidung von Schmauchspuren. | Brożek-Mucha, Z. 2015. Forensic Sci Int. 254: 51-8. PMID: 26188699
- Silicen Quantum Dots: Synthese, Spektroskopie und elektrochemische Studien. | Hu, P., et al. 2018. Langmuir. 34: 2834-2840. PMID: 29397732
- Strahlungsstimulierte Bildung zweidimensionaler Strukturen auf der Basis von Kalziumsilicid. | Zinovieva, AF., et al. 2022. Nanomaterials (Basel). 12: PMID: 36296813
- Chemische Bindungen und elektronische Zustände in Calciumsiliciden: Theorie und Vergleich mit Synchrotronstrahlungs-Photoemission. | Bisi, O., et al. 1989. Phys Rev B Condens Matter. 40: 10194-10209. PMID: 9991564
- Leere elektronische Zustände von Calciumsiliciden: Eine Untersuchung der inversen Photoemission im ultravioletten Photonenbereich. | Chemelli, C., et al. 1989. Phys Rev B Condens Matter. 40: 10210-10217. PMID: 9991565
- Silizium-Valenzzustände in Calciumsiliciden: Eine Si L2,3VV Auger-Linienform-Analyse. | Calliari, L., et al. 1990. Phys Rev B Condens Matter. 41: 7569-7575. PMID: 9993049
Bestellinformation
| Produkt | Katalog # | EINHEIT | Preis | ANZAHL | Favoriten | |
Calcium silicide, 250 g | sc-239471 | 250 g | $115.00 |
