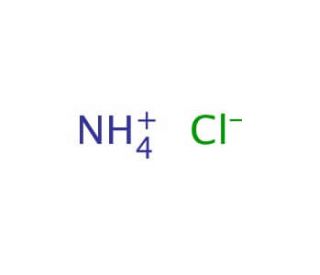
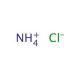
Ammonium Chloride (CAS 12125-02-9)
See product citations (4)
QUICK LINKS
Ammonium chloride, a white crystalline salt composed of ammonia and hydrochloric acid, is utilized extensively in various scientific research fields due to its unique chemical properties. In the realm of chemistry and environmental science, it is often employed as a nitrogen source in fertilizers, but its role extends significantly into analytical chemistry where it serves as a buffer in pH control. Ammonium chloride is particularly effective in maintaining a stable acidic environment, which is crucial during the titration processes where precise pH conditions are necessary to obtain accurate results. Its buffering capability is due to the equilibrium between ammonium ions and chloride ions, which can respond to changes in pH by absorbing or releasing hydrogen ions. This reaction is vital for experiments requiring consistent acid-base conditions. In addition to its buffering uses, ammonium chloride is also commonly used in the preparation of metal chlorides and in galvanizing processes. In research laboratories, it′s used in the lyophilization of cells, a technique for preserving materials by freezing and then drying them at low pressure. This chemical′s ability to dissociate into ammonia and hydrochloric acid upon heating makes it useful in thermal decomposition experiments, helping scientists study decomposition mechanisms and kinetics. Through these diverse applications, ammonium chloride proves itself as an invaluable tool in both industrial applications and scientific research, facilitating experiments that require precise control over chemical reactions and conditions.
Ammonium Chloride (CAS 12125-02-9) References
- Intracellular calcium responses to basic calcium phosphate crystals in fibroblasts. | Halverson, PB., et al. 1998. Osteoarthritis Cartilage. 6: 324-9. PMID: 10197167
- Mobilization, collection, and processing of peripheral blood stem cells in individuals with sickle cell trait. | Kang, EM., et al. 2002. Blood. 99: 850-5. PMID: 11806986
- Telomere length measurement by fluorescence in situ hybridization and flow cytometry: tips and pitfalls. | Baerlocher, GM., et al. 2002. Cytometry. 47: 89-99. PMID: 11813198
- N-Chlorotaurine and ammonium chloride: an antiseptic preparation with strong bactericidal activity. | Gottardi, W., et al. 2007. Int J Pharm. 335: 32-40. PMID: 17196346
- Removal of ammonium chloride generated by ammonia slip from the SNCR process in municipal solid waste incinerators. | Hwang, IH., et al. 2009. Chemosphere. 74: 1379-84. PMID: 19108871
- The effect of ammonium chloride on the multiplication of herpes simplex virus type 1 in Vero cells. | Koyama, AH. and Uchida, T. 1989. Virus Res. 13: 271-81. PMID: 2554609
- Ammonium chloride-catalyzed green multicomponent synthesis of dihydropyrazine and tetrahydrodiazepine derivatives 'on water'. | Shaabani, A., et al. 2019. Mol Divers. 23: 585-592. PMID: 30465252
- Ammonium chloride and ammonium sulfate for prevention of parturient paresis in dairy cows. | Oetzel, GR., et al. 1988. J Dairy Sci. 71: 3302-9. PMID: 3235732
- Reconstitution and crystallisation experiments with isolated split proteins from Bacillus stearothermophilus ribosomes. | Gewitz, HS., et al. 1987. Biochem Int. 15: 887-95. PMID: 3435552
- Mode of inhibition of diphtheria toxin by ammonium chloride. | Kim, K. and Groman, NB. 1965. J Bacteriol. 90: 1557-62. PMID: 5854584
- Purification of cord blood lymphocytes. | Ridings, J., et al. 1996. J Immunol Methods. 195: 43-8. PMID: 8814318
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium Chloride, 25 g | sc-202936 | 25 g | $39.00 | |||
Ammonium Chloride, 500 g | sc-202936A | 500 g | $55.00 | |||
Ammonium Chloride, 2.5 kg | sc-202936B | 2.5 kg | $150.00 |
