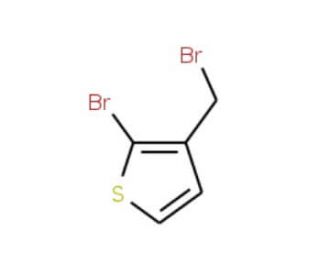
2-Bromo-3-(bromomethyl)thiophene (CAS 40032-76-6)
QUICK LINKS
2-Bromo-3-(bromomethyl)thiophene, also known as BBT, is an organic compound characterized by a heterocyclic ring containing sulfur. Moreover, it plays a role in investigating organic reactions and advancing catalyst development. Furthermore, 2-Bromo-3-(bromomethyl)thiophene facilitates the examination of molecular interactions like hydrogen bonding and pi-stacking. Although the exact mechanism of 2-Bromo-3-(bromomethyl)thiophene remains partially elusive, it is thought to function as a Lewis acid, forming complexes with Lewis bases. These complexes then partake in a series of reactions leading to the creation of novel compounds. Additionally, 2-Bromo-3-(bromomethyl)thiophene is believed to possess catalytic properties, thereby facilitating the formation of fresh compounds.
2-Bromo-3-(bromomethyl)thiophene (CAS 40032-76-6) References
- Design, synthesis, and biological evaluation of the N-diarylalkenyl-piperidinecarboxylic acid derivatives as GABA uptake inhibitors (I). | Zheng, J., et al. 2006. Bioorg Med Chem Lett. 16: 225-7. PMID: 16246548
- Synthesis of an organic-soluble π-conjugated [3]rotaxane via rotation of glucopyranose units in permethylated β-cyclodextrin. | Terao, J., et al. 2014. Beilstein J Org Chem. 10: 2800-8. PMID: 25550746
- Synthetic Utility of Arylmethylsulfones: Annulative π-Extension of Aromatics and Hetero-aromatics Involving Pd(0)-Catalyzed Heck Coupling Reactions. | Sankar, E., et al. 2017. J Org Chem. 82: 13583-13593. PMID: 29134811
- Synthesis and Thermal Investigations of Eleven-Membered Ring Systems Containing One of the Heavier Group 14 Element Atoms Si, Ge, and Sn. | Eleya, N., et al. 2020. Molecules. 25: PMID: 31936773
- Thiophene α-Chain-End-Functionalized Oligo(2-methyl-2-oxazoline) as Precursor Amphiphilic Macromonomer for Grafted Conjugated Oligomers/Polymers and as a Multifunctional Material with Relevant Properties for Biomedical Applications. | Bendrea, AD., et al. 2022. Int J Mol Sci. 23: PMID: 35886844
- Regioregular poly (thiophene-3-alkanoic acid) s: water soluble conducting polymers suitable for chromatic chemosensing in solution and solid state | Ewbank, P. C., Loewe, R. S., Zhai, L., Reddinger, J., Sauvé, G., & McCullough, R. D. 2004. Tetrahedron. 60(49): 11269-11275.
- Polythiophenes containing oligo (oxyethylene) side chains as a thin film on a ZnSe single crystal surface | van Beek, R., Jenneskens, L. W., Zdravkova, A. N., van der Eerden, J. P., & van Walree, C. A. 2005. Macromolecular Chemistry and Physics. 206(10): 1006-1014.
- Main chain polymeric metal complexes based on linkage fluorenevinylene or phenylenevinylene with thienyl (8‐hydro xyquinoline)–cadmium (II) complexes as dye sensitizer for dye‐sensitized solar cells | Jin, X., Yu, X., Zhang, W., Zhou, J., Tang, G., & Zhong, C. 2013. Journal of Applied Polymer Science. 129(6): 3104-3112.
- Bithiophene with Winding Vine-shaped Molecular Asymmetry. Preparation, Structural Characterization, and Enantioselective Synthesis | Toyomori, Y., Tsuji, S., Mitsuda, S., Okayama, Y., Ashida, S., Mori, A.,.. & Ogasawara, M. 2016. Bulletin of the Chemical Society of Japan. 89(12): 1480-1486.
- Fine-tuning the solid-state ordering and thermoelectric performance of regioregular P3HT analogues by sequential oxygen-substitution of carbon atoms along the alkyl side chains | Chen, L., Liu, W., Yan, Y., Su, X., Xiao, S., Lu, X.,.. & Tang, X. 2019. Journal of Materials Chemistry C. 7(8): 2333-2344.
- A study on regulating the conjugate position of NLO chromophores for reducing the dipole moment and enhancing the electro-optic activities of organic materials | Zhang, H., Tian, Y., Bo, S., Xiao, L., Ao, Y., Zhang, J., & Li, M. 2020. ournal of Materials Chemistry C. 8(4): 1380-1390.
- First Report of Chenodeoxycholic Acid–Substituted Dyes Improving the Dye Monolayer Quality in Dye-Sensitized Solar Cells | Buene, A. F., Almenningen, D. M., Hagfeldt, A., Gautun, O. R., & Hoff, B. H. 2020. Solar Rrl. 4(4): 1900569.
- A review: methodologies for the synthesis of anthra [2, 3-b] thiophene and naphtho [2, 3-b: 6, 7-b'] dithiophene fragments for organic semiconductor materials | Mustafa, A., & Abid, M. A. 2022. Tetrahedron Letters. 113: 154258.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Bromo-3-(bromomethyl)thiophene, 5 g | sc-254161 | 5 g | $183.00 |
