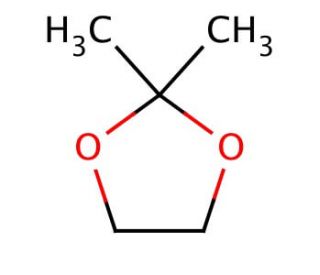
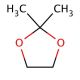
2,2-Dimethyl-1,3-dioxolane (CAS 2916-31-6)
QUICK LINKS
2,2-Dimethyl-1,3-dioxolane is an organic compound with applications in various research domains. Its dioxolane ring structure makes it a valuable solvent in organic synthesis, where it is used to dissolve both polar and non-polar substances, allowing for a wide range of chemical reactions. In the field of green chemistry, it is studied for its potential as a bio-based solvent, aiming to replace more toxic or environmentally harmful alternatives. Additionally, this compound is investigated for its use as a stabilizing agent in the production of chlorinated solvents, where it can help in reducing the formation of acid during storage. Researchers also examine the ring-opening polymerization of 2,2-dimethyl-1,3-dioxolane as a route to produce polyacetals, which are important in creating biodegradable plastics.
2,2-Dimethyl-1,3-dioxolane (CAS 2916-31-6) References
- Synthesis, swelling behavior, and biocompatibility of novel physically cross-linked polyurethane-block-poly(glycerol methacrylate) hydrogels. | Mequanint, K., et al. 2006. Biomacromolecules. 7: 883-91. PMID: 16529427
- Rapid analysis of tile industry gaseous emissions by ion mobility spectrometry and comparison with solid phase micro-extraction/gas chromatography/mass spectrometry. | Pozzi, R., et al. 2006. J Environ Monit. 8: 1219-26. PMID: 17133278
- Pyrene magic: chiroptical enciphering and deciphering 1,3-dioxolane bearing two wirepullings to drive two remote pyrenes. | Amako, T., et al. 2015. Chem Commun (Camb). 51: 8237-40. PMID: 25820177
- Application of acetone acetals as water scavengers and derivatization agents prior to the gas chromatographic analysis of polar residual solvents in aqueous samples. | van Boxtel, N., et al. 2015. J Chromatogr A. 1425: 62-72. PMID: 26614172
- Synthesis, structure and pyrolysis of stabilised phosphonium ylides containing saturated oxygen heterocycles. | Aitken, RA., et al. 2016. Org Biomol Chem. 14: 1794-804. PMID: 26751733
- Design, synthesis and evaluation of 2,2-dimethyl-1,3-dioxolane derivatives as human rhinovirus 3C protease inhibitors. | Zhang, Q., et al. 2017. Bioorg Med Chem Lett. 27: 4061-4065. PMID: 28778471
- Chemical constituents of Antidesma bunius aerial parts and the anti-AGEs activity of selected compounds. | Nguyen-Ngoc, H., et al. 2022. Phytochemistry. 202: 113300. PMID: 35798090
- Potential Role of Low-Molecular-Weight Dioxolanes as Adjuvants for Glyphosate-Based Herbicides Using Photosystem II as an Early Post-Treatment Determinant. | Szwajczak, E., et al. 2023. Cells. 12: PMID: 36899913
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2-Dimethyl-1,3-dioxolane, 25 g | sc-238228 | 25 g | $117.00 |
