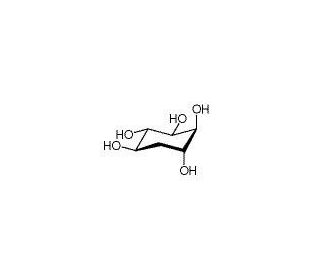
(+)-proto-Quercitol (CAS 488-73-3)
QUICK LINKS
(+)-proto-Quercitol, a naturally occurring polyol found in various plants, has gained attention in scientific research for its antioxidant properties and potential health benefits. Studies have explored its mechanism of action, revealing its ability to scavenge free radicals and reduce oxidative stress-induced damage in cells and tissues. This compound has been investigated for its role in mitigating inflammation and modulating immune responses, suggesting its potential applications in the prevention and management of chronic inflammatory diseases. Additionally, research has focused on elucidating the bioavailability and metabolism of (+)-proto-Quercitol, providing insights into its pharmacokinetic properties and potential interactions with other dietary components. Furthermore, (+)-proto-Quercitol has been studied for its effects on cellular signaling pathways, particularly those involved in regulating cell proliferation and apoptosis. By unraveling the mechanisms underlying the biological activities of (+)-proto-Quercitol, researchers aim to uncover its potential applications and explore its use as a natural antioxidant and nutraceutical. Overall, (+)-proto-Quercitol continues to be a subject of scientific interest, driving advancements in antioxidant research and contributing to the exploration of natural compounds for various research applications.
(+)-proto-Quercitol (CAS 488-73-3) References
- Transformation of quercitols into 4-methylenecyclohex-5-ene-1,2,3-triol derivatives, precursors for the chemical chaperones N-octyl-4-epi-β-valienamine (NOEV) and N-octyl-β-valienamine (NOV). | Kuno, S., et al. 2011. Bioorg Med Chem Lett. 21: 7189-92. PMID: 22001090
- Concise synthesis of (+)-conduritol F and inositol analogues from naturally available (+)-proto-quercitol and their glucosidase inhibitory activity. | Worawalai, W., et al. 2012. Bioorg Med Chem Lett. 22: 1538-40. PMID: 22290077
- Concise syntheses of potent chaperone drug candidates, N-octyl-4-epi-β-valinenamine (NOEV) and its 6-deoxy derivative, from (+)-proto-quercitol. | Kuno, S., et al. 2013. Carbohydr Res. 368: 8-15. PMID: 23314299
- Quercitylcinnamates, a new series of antidiabetic bioconjugates possessing α-glucosidase inhibition and antioxidant. | Rattanangkool, E., et al. 2013. Eur J Med Chem. 66: 296-304. PMID: 23811091
- Amine-linked diquercitols as new α-glucosidase inhibitors. | Worawalai, W., et al. 2014. Bioorg Med Chem Lett. 24: 5530-3. PMID: 25451999
- Voglibose-inspired synthesis of new potent α-glucosidase inhibitors N-1,3-dihydroxypropylaminocyclitols. | Worawalai, W., et al. 2016. Carbohydr Res. 429: 155-62. PMID: 27140506
- Efficient and Highly Stereoselective Syntheses of (+)-proto-Quercitol and (-)-gala-Quercitol Starting from the Naturally Abundant (-)-Shikimic Acid. | Zhu, XL., et al. 2020. ACS Omega. 5: 1813-1821. PMID: 32039317
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(+)-proto-Quercitol, 100 mg | sc-296145 | 100 mg | $462.00 |
