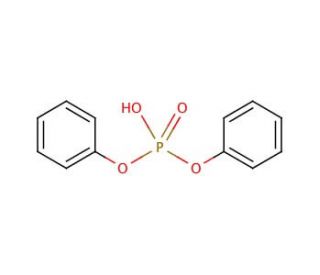
Diphenyl phosphate (CAS 838-85-7)
QUICK LINKS
Diphenyl phosphate is a chemical compound that functions as a flame retardant in various materials. It acts by forming a protective char layer on the surface of the material when exposed to fire or high temperatures. This char layer acts as a barrier, reducing the flammability and slowing down the spread of fire. At a molecular level, diphenyl phosphate undergoes a series of chemical reactions when exposed to heat, leading to the formation of the char layer. This mechanism of action effectively inhibits the combustion process and contributes to the overall fire resistance of the material. In experimental applications, diphenyl phosphate is utilized to study the impact of flame retardants on the flammability and fire safety of different materials.
Diphenyl phosphate (CAS 838-85-7) References
- Analysis of the flame retardant metabolites bis(1,3-dichloro-2-propyl) phosphate (BDCPP) and diphenyl phosphate (DPP) in urine using liquid chromatography-tandem mass spectrometry. | Cooper, EM., et al. 2011. Anal Bioanal Chem. 401: 2123-32. PMID: 21830137
- Unique and highly selective anticytomegalovirus activities of artemisinin-derived dimer diphenyl phosphate stem from combination of dimer unit and a diphenyl phosphate moiety. | He, R., et al. 2013. Antimicrob Agents Chemother. 57: 4208-14. PMID: 23774439
- Quantification of three chlorinated dialkyl phosphates, diphenyl phosphate, 2,3,4,5-tetrabromobenzoic acid, and four other organophosphates in human urine by solid phase extraction-high performance liquid chromatography-tandem mass spectrometry. | Jayatilaka, NK., et al. 2017. Anal Bioanal Chem. 409: 1323-1332. PMID: 27838756
- Associations between urinary diphenyl phosphate and thyroid function. | Preston, EV., et al. 2017. Environ Int. 101: 158-164. PMID: 28162782
- Presence of diphenyl phosphate and aryl-phosphate flame retardants in indoor dust from different microenvironments in Spain and the Netherlands and estimation of human exposure. | Björnsdotter, MK., et al. 2018. Environ Int. 112: 59-67. PMID: 29268159
- Neonatal triphenyl phosphate and its metabolite diphenyl phosphate exposure induce sex- and dose-dependent metabolic disruptions in adult mice. | Wang, D., et al. 2018. Environ Pollut. 237: 10-17. PMID: 29466770
- Assessment of impacts of diphenyl phosphate on groundwater and near-surface environments: Sorption and toxicity. | Funk, SP., et al. 2019. J Contam Hydrol. 221: 50-57. PMID: 30642690
- Diphenyl Phosphate-Induced Toxicity During Embryonic Development. | Mitchell, CA., et al. 2019. Environ Sci Technol. 53: 3908-3916. PMID: 30864794
- Cd-Based Metal-Organic Framework Containing Uncoordinated Carbonyl Groups as Lanthanide Postsynthetic Modification Sites and Chemical Sensing of Diphenyl Phosphate as a Flame-Retardant Biomarker. | Qu, XL. and Yan, B. 2020. Inorg Chem. 59: 15088-15100. PMID: 33006286
- Life Cycle Exposure to Environmentally Relevant Concentrations of Diphenyl Phosphate (DPhP) Inhibits Growth and Energy Metabolism of Zebrafish in a Sex-Specific Manner. | Chen, Q., et al. 2021. Environ Sci Technol. 55: 13122-13131. PMID: 34523920
- Triphenyl phosphate proved more potent than its metabolite diphenyl phosphate in inducing hepatic insulin resistance through endoplasmic reticulum stress. | Yue, J., et al. 2023. Environ Int. 172: 107749. PMID: 36680801
- The aomogeneous and heterogeneous oxidation of organophosphate esters (OPEs) in the atmosphere: Take diphenyl phosphate (DPhP) as an example. | Huo, Y., et al. 2023. Environ Pollut. 324: 121395. PMID: 36871750
- Diphenyl Phosphate as an Efficient Cationic Organocatalyst for Controlled/Living Ring-Opening Polymerization of δ-Valerolactone and ε-Caprolactone | Kosuke Makiguchi, Toshifumi Satoh, and Toyoji Kakuchi*. 2011,. Macromolecules. 44, 7,: 1999–2005.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diphenyl phosphate, 5 g | sc-239798 | 5 g | $37.00 |
