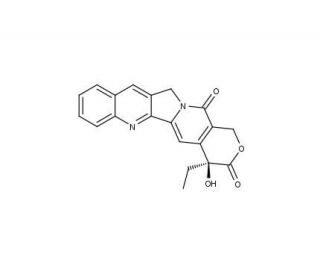
Camptothecin (CAS 7689-03-4)
See product citations (21)
QUICK LINKS
Camptothecin is an alkaloid compound originally found in the Chinese tree Camptotheca acuminata. In research applications, camptothecin is valued for its ability to inhibit DNA topoisomerase I, an enzyme critical for DNA replication and transcription. By stabilizing the transient break in the double-stranded DNA that topoisomerase I creates, camptothecin induces DNA damage and ultimately cell death, which is particularly relevant for studies on cell proliferation and viability. This characteristic has made it a focal point in the study of mechanisms that cancer cells use to proliferate and survive, as well as in the exploration of potential strategies for inducing controlled cell death. Its role in inducing apoptosis through the DNA damage response pathway further contributes to understanding the intricate balance between cell survival and death. Research with camptothecin also extends to the field of molecular biology, providing insights into the dynamics of DNA replication and the cellular response to genotoxic stress.
Camptothecin (CAS 7689-03-4) References
- Body distribution in mice of intravenously injected camptothecin solid lipid nanoparticles and targeting effect on brain. | Yang, SC., et al. 1999. J Control Release. 59: 299-307. PMID: 10332062
- Distribution of camptothecin after delivery as a liposome aerosol or following intramuscular injection in mice. | Koshkina, NV., et al. 1999. Cancer Chemother Pharmacol. 44: 187-92. PMID: 10453719
- Camptothecin suppresses nitric oxide biosynthesis in RAW 264.7 macrophages. | Chiou, WF., et al. 2001. Life Sci. 69: 625-35. PMID: 11476184
- Camptothecin and its analogues: a review on their chemotherapeutic potential. | Sriram, D., et al. 2005. Nat Prod Res. 19: 393-412. PMID: 15938148
- Irreversible trapping of the DNA-topoisomerase I covalent complex. Affinity labeling of the camptothecin binding site. | Hertzberg, RP., et al. 1990. J Biol Chem. 265: 19287-95. PMID: 2172250
- Metabolism and pharmacokinetics of the camptothecin analogue CPT-11 in the mouse. | Kaneda, N., et al. 1990. Cancer Res. 50: 1715-20. PMID: 2306725
- Sustainable production of camptothecin from an Alternaria sp. isolated from Nothapodytes nimmoniana. | Mohinudeen, IAHK., et al. 2021. Sci Rep. 11: 1478. PMID: 33446714
- Studies on the antitumor activity, mechanism of action, and cell cycle effects of camptothecin. | Gallo, RC., et al. 1971. J Natl Cancer Inst. 46: 789-95. PMID: 4995657
- Differential stabilization of eukaryotic DNA topoisomerase I cleavable complexes by camptothecin derivatives. | Tanizawa, A., et al. 1995. Biochemistry. 34: 7200-6. PMID: 7766631
- The current status of camptothecin analogues as antitumor agents. | Slichenmyer, WJ., et al. 1993. J Natl Cancer Inst. 85: 271-91. PMID: 8381186
- The anti-cancer drug camptothecin inhibits elongation but stimulates initiation of RNA polymerase II transcription. | Ljungman, M. and Hanawalt, PC. 1996. Carcinogenesis. 17: 31-5. PMID: 8565133
- Chemotherapeutic agent CPT-11 induces the new expression of the apoptosis initiator to the cytoplasm. | Suzuki, A. and Kato, M. 1996. Exp Cell Res. 227: 154-9. PMID: 8806462
- Camptothecin-induced apoptosis in p53-null human leukemia HL60 cells and their isolated nuclei: effects of the protease inhibitors Z-VAD-fmk and dichloroisocoumarin suggest an involvement of both caspases and serine proteases. | Shimizu, T. and Pommier, Y. 1997. Leukemia. 11: 1238-44. PMID: 9264376
- Mechanism of action of eukaryotic DNA topoisomerase I and drugs targeted to the enzyme. | Pommier, Y., et al. 1998. Biochim Biophys Acta. 1400: 83-105. PMID: 9748515
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Camptothecin, 50 mg | sc-200871 | 50 mg | $58.00 | |||
Camptothecin, 100 mg | sc-200871B | 100 mg | $94.00 | |||
Camptothecin, 250 mg | sc-200871A | 250 mg | $186.00 |
